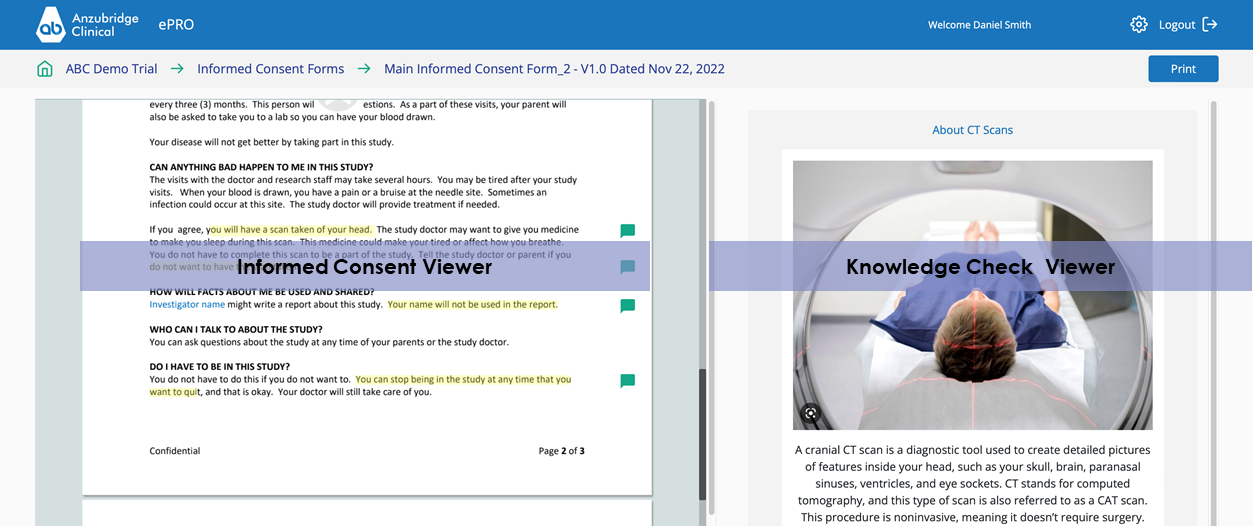
e-Consent
KNOWLEDGE CHECKS
Add Knowledge Checks to your Informed Consent using the Anzubridge® e-Consent Builder.
- Add an introductory video to your Informed Consent document.
- Use our Help Template Designer to create supplemental content.
- Link that content to specific text in your document.
- Questions and answers
- Text
- Images
- Image list
- Image and text
- Videos
- Audio file
- Add post-review questions (optional) to your Informed Consent document to validate that the study participant has reviewed the document and the associated Knowledge Checks
Study participants can view the Informed Consent, review the Knowledge Checks, and e-sign the document, all on one screen.
Q&As
The FDA does not require the use of electronic informed consents (e-Consent) with knowledge checks for all clinical trials. However, the FDA recognizes the benefits of using eConsent, including increased patient engagement and understanding of the trial, and improved accuracy and efficiency of the consent process.
The FDA has issued guidance documents on the use of e-Consent in clinical trials, which outline the requirements and considerations for implementing eConsent in trials. The guidance encourages the use of e-Consent with knowledge checks as a way to enhance patient understanding and engagement in the consent process.
While the use of e-Consent is not required by the FDA, it is important for companies to consider its use in clinical trials, particularly as technology advances and patient expectations for electronic engagement continue to evolve. Additionally, the use of eConsent may be required by specific regulatory agencies in certain countries, so it’s important to review local regulations and requirements.
The source document for FDA guidance on electronic informed consents with knowledge checks is the “Guidance for Industry: Use of Electronic Informed Consent in Clinical Investigations.”
This guidance document provides recommendations for the use of electronic informed consent in clinical trials, including the use of knowledge checks to ensure that subjects understand the information presented in the informed consent process. The guidance outlines the importance of using clear and concise language in electronic informed consents, as well as the need for secure and regulated storage of these data.
This document can be found on the FDA’s website at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/use-electronic-informed-consent-clinical-investigations-questions-and-answers